Why can't I search by lot number?
Subscribe to Receive Our Weekly Call To Action by Email
There are surely bad batches of vaccine out there. We know this, we have known it for a long time. It’s the reason they now break lots up and distribute them across the country.[1] Whether you can prove this with the limited VAERS data that we have access to (we, as opposed to what the FDA, CDC and Pharma have) is another story. The jury is out in our opinion on trying to make the case. We believe there are 3 key questions that need to be addressed and remain skeptical that it’s possible without the three letter agencies to actually address them.
First, some background. The lot number data is very incomplete. It’s clear looking at the entries that a large number of lot numbers are missing, mistaken, and just plain incoherent. Some say things like “I can't read the number”. Some have dates in them. Some are just a few letters or numbers. They cannot be used without cleaning. Cleaning is arduous and difficult to do by hand and subject to human error. While it is possible to script to clean the data, the scripts will never catch all the problems unless you already had a list of lot numbers to compare to, which only the 3 letter agencies have.
Additionally, in some cases there are two or even three doses/lot numbers listed without an easy way to determine which lot caused the problem. If we are then counting up injuries for lot numbers do we count each dose? The first? The last? And if we choose how do we know in the case of multiple lots which caused the problem or was it cumulative? This is not a little thing. In the end we likely just throw out the multi-batch reports and just use the single. But even then it’s confusing. You can even see some reporters trying to clarify by putting “first dose” or “second dose” next to the lot number.
Finally, it’s important to keep in mind that VAERS itself is a database of possible injury in a larger set of all given vaccines. We can postulate the denominator or make comparisons. HOWEVER, the lot numbers are a subset of a subset of the total data set. It is a subset of damage. All the low number batches reports are mostly mistakes. How many batches are out there that produced no VAERS reports? We have no idea.
Okay and that was just the background. Here are the questions that need to be answered.
What are the batch sizes?
We have no idea. Some can speculate but it cannot be assumed.
What is the sum totality of batches extant? How many batches are in the market?
Again we have no idea, only FDA and Pharma can answer this question with certitude.
How do we deal with the backfill?
There is is a time frame issue with the data, all the data, that cannot be ignored. We call it backfilling. Basically older batches will have a larger number of reports because VAERS enters reports from all time frames at their discretion and we see this in the data. This is a major confounding problem when looking at the level of "injury" based upon the number of reports in a given batch.
Below are 2 charts showing this—one by RECVDATE and one by VAX_DATE. The X axis is the month the VAERS report was received or the month the vaccine was received, the Y axis is the number of reports made. Each color is a distinct batch number. We removed the lot numbers that have fewer than 5 reports as these tend to be the worst, most broken data. This data is from Pfizer and US only, with all unknown lots removed. Is this distribution because the batch is more toxic or is this distribution because the VAERS system is backfilling early reports and these lot numbers appear earlier in the data?
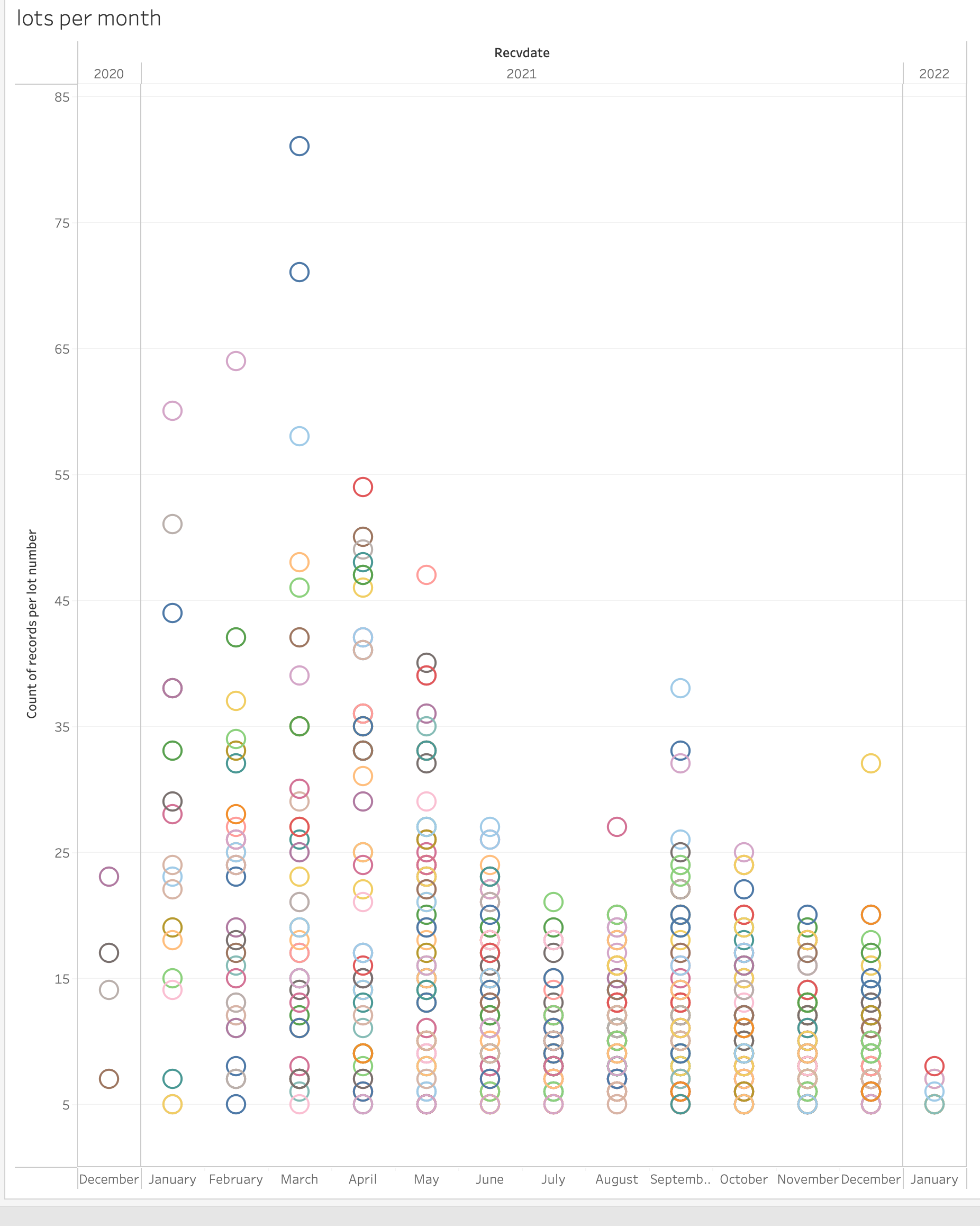
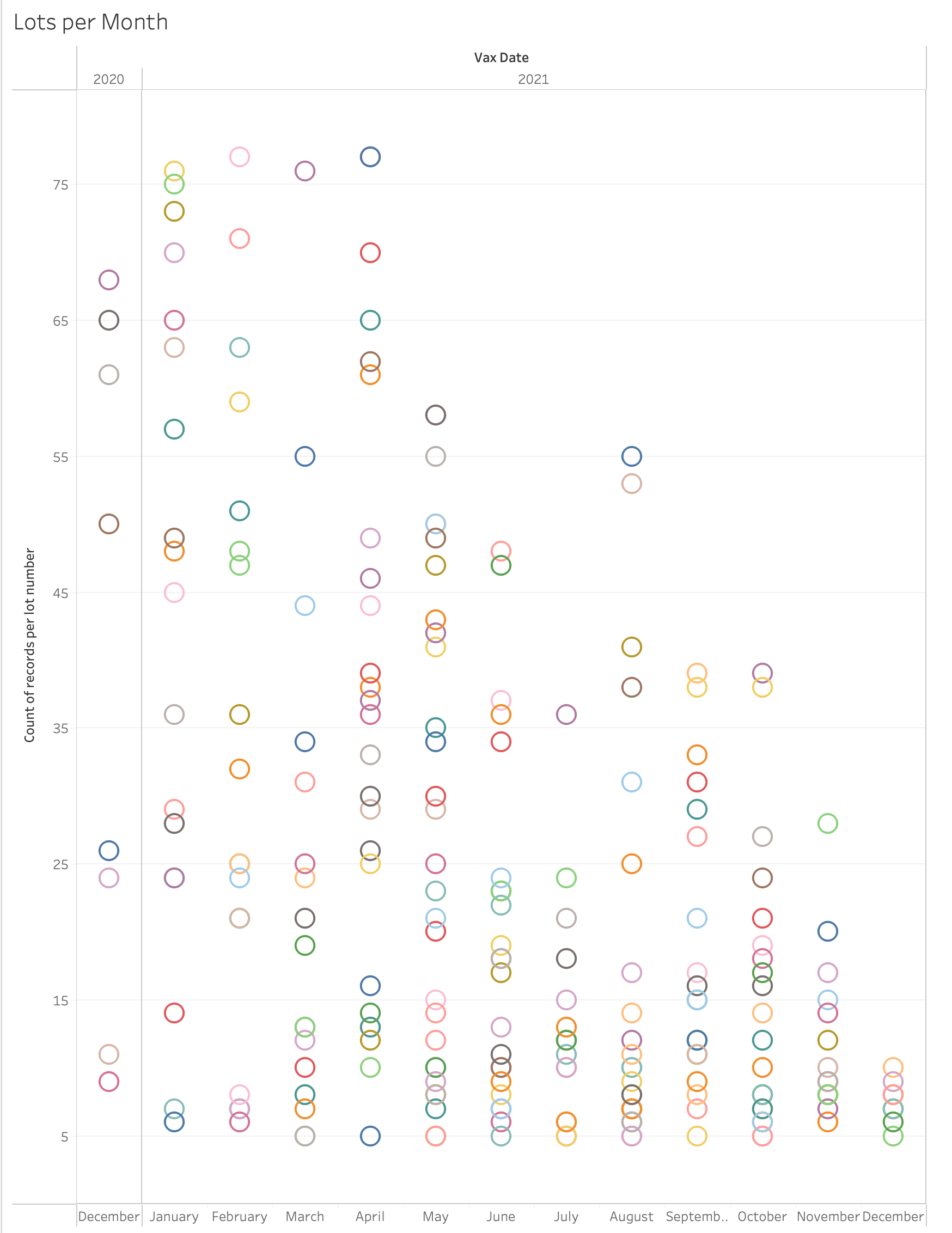
Matthew Crawford also addresses this issue, along with a few others not covered here, in his Substack article: https://roundingtheearth.substack.com/p/understanding-batch-lot-toxicity. He hypothesizes that the backfill also correlates with age. "Jessica [Rose] produced a nice plot showing that the potentially hot lots usually happened earlier in the vaccination campaign where we would assume there is high correlation with age of the dose recipient." Dr. Rose's plot is below, here is her article about this topic: https://jessicar.substack.com/p/getting-to-the-bottom-of-potential.
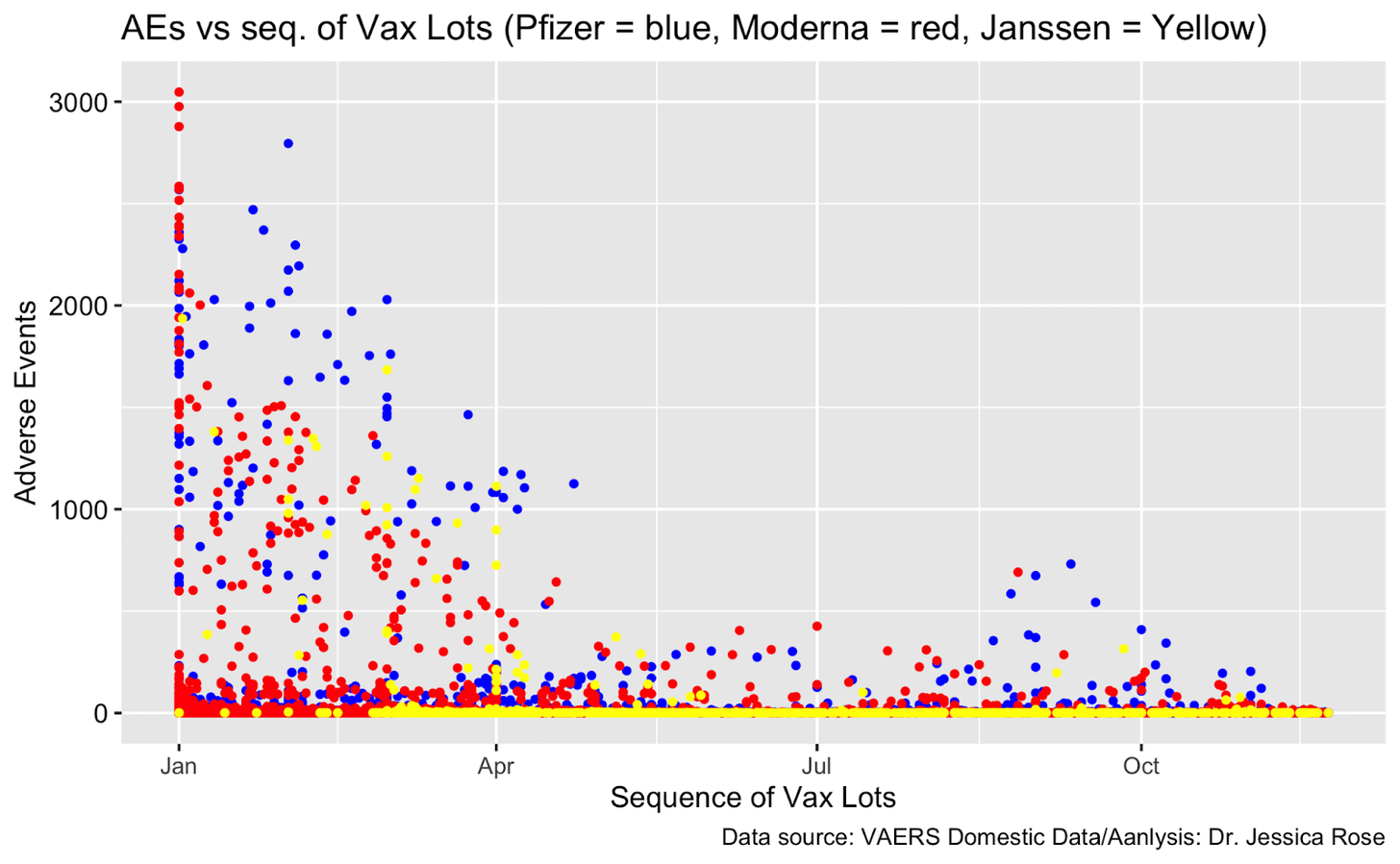
So currently while we think there are assuredly bad batches, we do not believe that VAERS alone can prove it.
1. https://www.ageofautism.com/2008/08/by-dan-olmsted.html